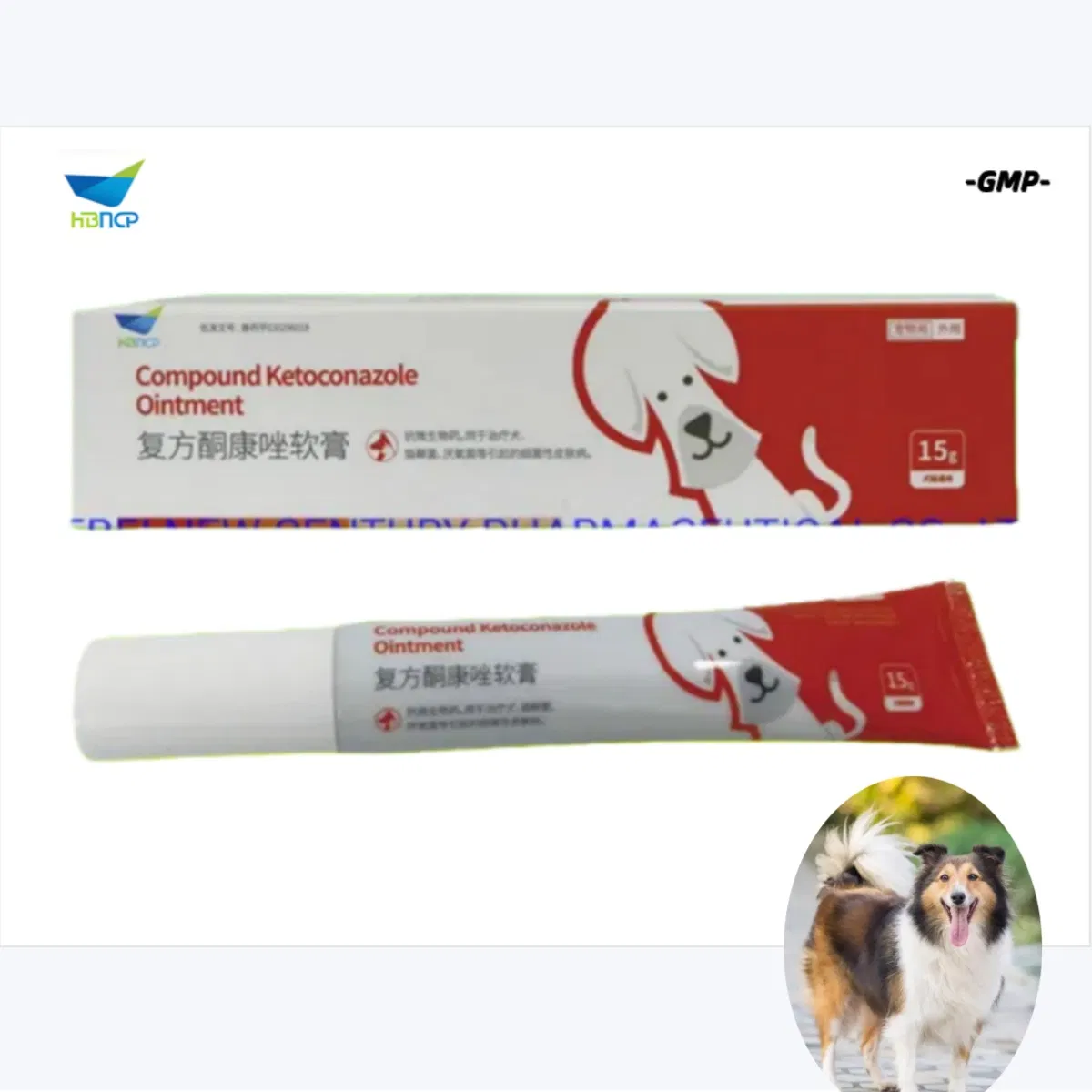
Product Description
Compound Ketoconazole Ointment For Pets Animals Skin Fungi Infection
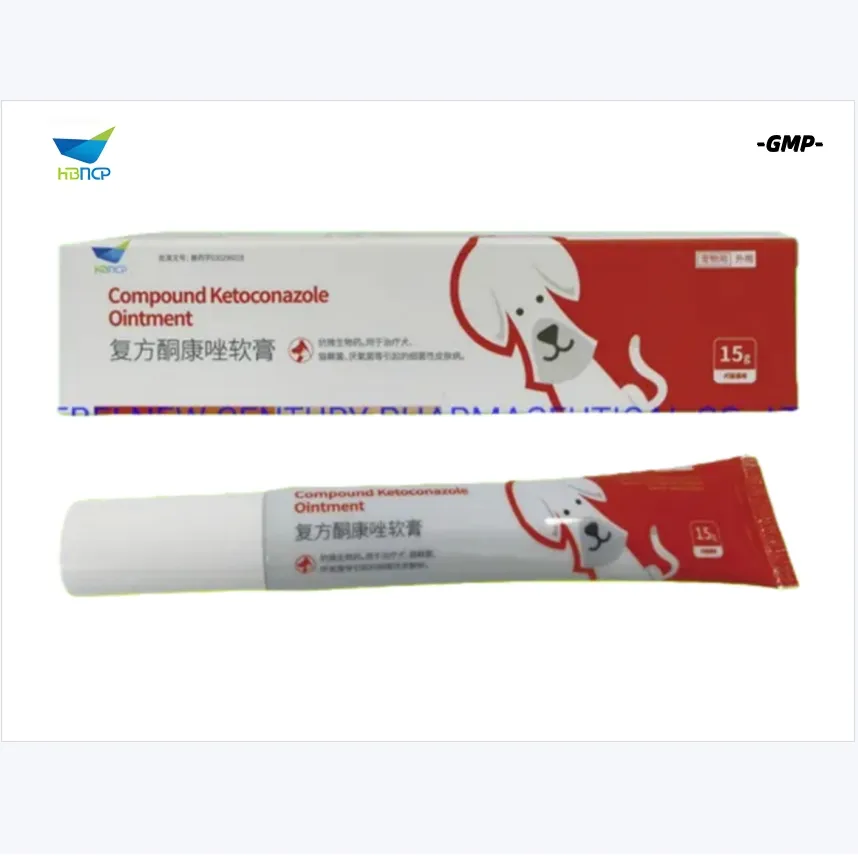
Product Specifications & Composition:
| Ketoconazole | 1.0% |
| Menthol crystals | 1.0% |
Advanced Formula:
| Ketoconazole | 1.0% |
| Clobetasol propionate | 0.025% |
| Neomycin sulfate | 5000iu |
| Excipient | q.s 100% |
Ketoconazole + Clobetasol + Neomycin Cream is used for severe inflammatory skin conditions such as eczema, dermatitis, or psoriasis that are infected. It reduces skin inflammation (redness, swelling, itching) and treats serious fungal or yeast infections.
Manufacturing Excellence

- ★ 18 Years GMP Certificated
- ★ Comprehensive 14 Production Lines
- ★ Stable Quality & Competitive Price
- ★ 100% Timely Delivery
- ★ Passed GMP inspection in multiple countries
Quality Control & Laboratory
The facility utilizes high-performance liquid chromatographs, UV-visible spectrophotometers, and gas chromatographs to ensure every batch meets required pharmacopoeia standards.
Exhibitions & Global Reach
Certifications
Frequently Asked Questions
Q1: Does the product have the necessary qualifications for export?
A: Yes, our production holds GMP certification. We provide COA, Free Sale Certificates (FSC), and Certificates of Origin to meet importing country requirements.
Q2: What quality standards do the products comply with?
A: All products are strictly produced according to the standards of the Chinese Veterinary Pharmacopoeia and international GMP norms.
Q3: What are the primary uses of Ketoconazole Ointment?
A: It is used for treating fungal infections, yeast infections (like candidiasis), and inflammatory skin conditions such as eczema or dermatitis in animals.
Q4: How should this ointment be stored?
A: It should be stored in a light-proof environment to maintain the stability of the active ingredients.
Q5: What is the production capacity for this product?
A: Our current production capacity is approximately 20,000 tubes per day across multiple advanced production lines.
Q6: Are the production facilities inspected internationally?
A: Yes, the facilities have successfully passed GMP inspections in various countries including Ethiopia, Sudan, and Uganda.