ℹ️
Basic Info
Certification
CE, FDA, GMP, ISO
Control System
Fully Automatic
Treatment Process
Filtration, Ultrafiltration
Core Components Warranty
1 Year
Tank Material
Stainless Steel 316L
📋
Product Description
Biologics Vaccination Solution New drug R&D Formulation Vessel Mixing Tank System
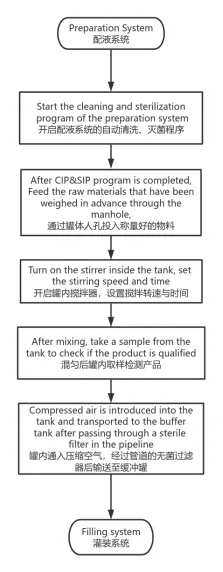
The preparation system is specialized pharmaceutical equipment used to mix and dispense liquids. It integrates Active Pharmaceutical Ingredients (API) with solvents like water for injection, followed by stirring, heating, sterilization, and filtration to achieve precise concentrations.
🛠️ System Classifications
- General Preparation: For small molecule chemical injections and lyophilized powders.
- Complex Preparation: For suspensions, emulsions, liposomes, and microspheres.
- Biological Preparation: For antibodies, vaccines, blood products, and recombinant proteins.
- CIP Station: Integrated on-line cleaning for tanks and pipelines.
📊
Product Parameters
| Name | Detailed Parameters |
|---|
| Working Volume | 2 ~ 15000L |
| Stirring Speed Control | 0 ~ 450rpm (±3.0%) |
| Temperature Range | -10 ~ 150ºC (±0.2ºC) |
| Pressure Control | -0.01Mpa ~ 0.06Mpa (±0.01MPa) |
| Weighing Accuracy | 3‰ |
| Power Supply | 380V 50Hz |
| Protection Level | IP65 |
🖼️
Technical Showcase

❓
Frequently Asked Questions
What materials are used for the mixing tanks?
The system utilizes high-grade Stainless Steel 316L, ensuring excellent corrosion resistance and hygiene for pharmaceutical use.
What is the range of working volumes available?
We offer a wide range of working volumes from small-scale 2L units up to large industrial 15,000L systems.
Is the system compatible with CIP/SIP processes?
Yes, the system is designed to support Cleaning-In-Place (CIP) and can be integrated with sterilization protocols to ensure aseptic conditions.
What kind of temperature control accuracy can be expected?
The system provides highly precise temperature control with an accuracy of ±0.2ºC within a range of -10 to 150ºC.
Can this system be used for vaccine production?
Absolutely. The Biological Preparation System classification is specifically designed for vaccines, antibodies, and recombinant protein production.
What are the electrical requirements?
The standard power supply is 380V 50Hz, but other voltage requirements can be accommodated via a transformer.